NEWS CENTER
Selective reaction of carbonyl group
Release time:
2023-06-13 17:26
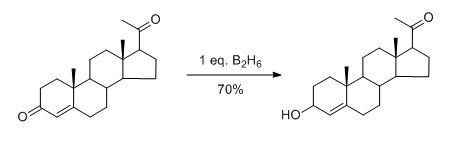
Carbonyl is another kind of important functional group, which is often used to study selective reactions. For example, due to the different electron delocalization of α, β - unsaturated ketone and saturated ketone, nucleophilic reductant h can attack the saturated ketone negatively and selectively, while electrophilic reagent B2H6 can react with the unsaturated ketone selectively.
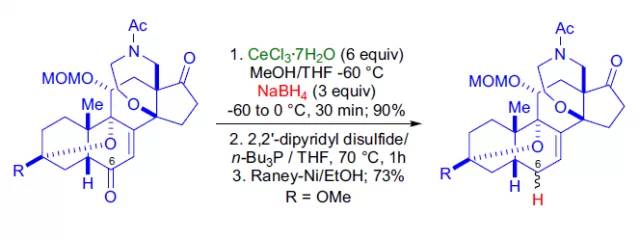
Luche et al found that the combination of NaBH4 and cecl3.6h2o can selectively reduce α, β - unsaturated aldehydes and ketones, which is widely used in the synthesis of complex systems.
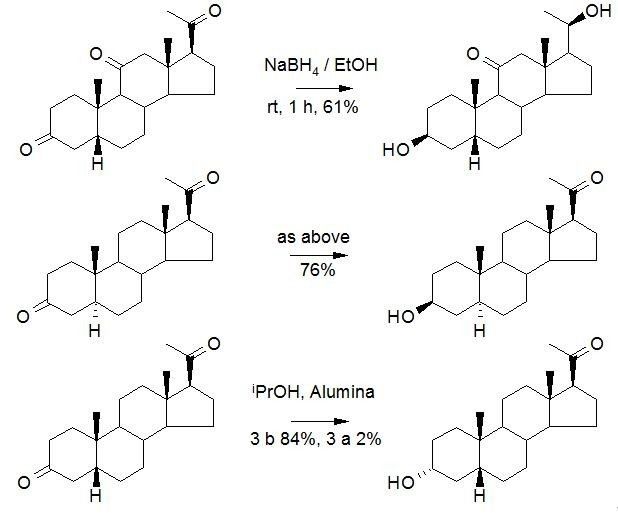
In the same way, the difference of some chemical environments can also be used for selective reactions between saturated ketones. As mentioned above, the framework of steroids is a rigid environment with large regional position difference, so it is often used for the selective reaction of functional groups, and carbonyl is no exception.
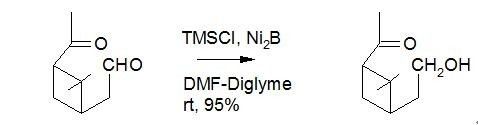
In addition, the reaction between aldehydes and ketones can also be selected. In general, the activity of aldehyde group is slightly higher, and the reaction usually takes place preferentially; but in some cases, the opposite selectivity can also be obtained, that is, ketone can also take place preferentially.
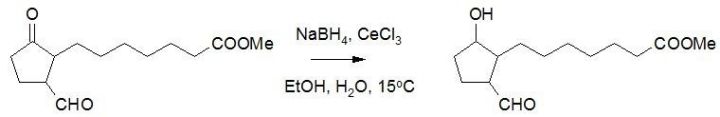
Service Hotline: South China region15124358911/East China region13634423569
If you have any questions, please feel free to contact us


